Filter News
Area of Research
- (-) Materials (86)
- Advanced Manufacturing (9)
- Biological Systems (4)
- Biology and Environment (4)
- Biology and Soft Matter (3)
- Building Technologies (4)
- Chemical and Engineering Materials (2)
- Chemistry and Physics at Interfaces (5)
- Clean Energy (79)
- Climate and Environmental Systems (6)
- Computational Biology (1)
- Computational Chemistry (4)
- Computational Engineering (2)
- Computer Science (9)
- Earth Sciences (1)
- Energy Frontier Research Centers (6)
- Fuel Cycle Science and Technology (1)
- Functional Materials for Energy (8)
- Fusion Energy (6)
- Geographic Information Science and Technology (1)
- Materials for Computing (7)
- Materials Synthesis from Atoms to Systems (8)
- Materials Under Extremes (5)
- National Security (5)
- Neutron Data Analysis and Visualization (2)
- Neutron Science (29)
- Nuclear Science and Technology (16)
- Nuclear Systems Technology (1)
- Quantum Condensed Matter (2)
- Quantum information Science (3)
- Renewable Energy (1)
- Sensors and Controls (1)
- Supercomputing (46)
- Transportation Systems (4)
News Topics
- 3-D Printing/Advanced Manufacturing (5)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Bioenergy (2)
- Clean Water (2)
- Composites (1)
- Computer Science (3)
- Cybersecurity (1)
- Energy Storage (4)
- Environment (5)
- Fusion (2)
- Isotopes (1)
- Materials Science (19)
- Microscopy (5)
- Molten Salt (1)
- Nanotechnology (6)
- Neutron Science (5)
- Nuclear Energy (6)
- Physics (2)
- Polymers (2)
- Quantum Science (2)
- Sustainable Energy (5)
- Transportation (5)
Media Contacts
Scientists at the Department of Energy’s Oak Ridge National Laboratory (ORNL) have developed a process that could remove CO2 from coal-burning power plant emissions in a way that is similar to how soda lime works in scuba diving rebreathers. Their research, published January 31 in...

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.

Jon Poplawsky, a materials scientist at the Department of Energy’s Oak Ridge National Laboratory, develops and links advanced characterization techniques that improve our ability to see and understand atomic-scale features of diverse materials
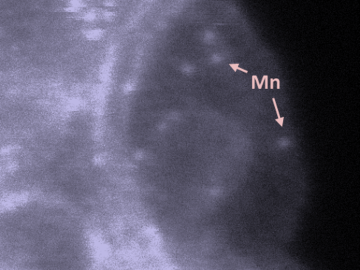
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.


Scientists at the Department of Energy’s Oak Ridge National Laboratory have made the first direct observations of a one-dimensional boundary separating two different, atom-thin materials, enabling studies of long-theorized phenomena at these interfaces. Theorists h...
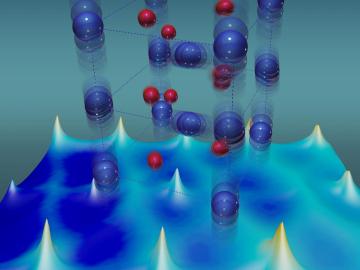
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.