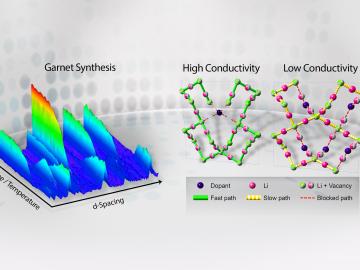
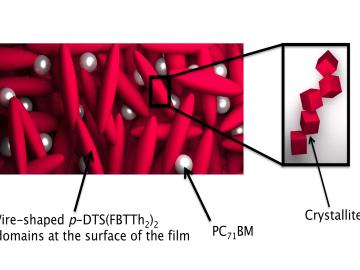
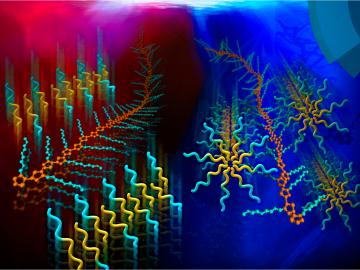
Filter News
Area of Research
- (-) Neutron Science (33)
- Advanced Manufacturing (9)
- Biological Systems (4)
- Biology and Environment (4)
- Biology and Soft Matter (1)
- Building Technologies (3)
- Chemical and Engineering Materials (2)
- Chemistry and Physics at Interfaces (6)
- Clean Energy (101)
- Climate and Environmental Systems (2)
- Computational Chemistry (1)
- Computational Engineering (1)
- Computer Science (8)
- Energy Frontier Research Centers (7)
- Functional Materials for Energy (6)
- Fusion Energy (7)
- Geographic Information Science and Technology (2)
- Isotope Development and Production (1)
- Materials (94)
- Materials Synthesis from Atoms to Systems (5)
- Materials Under Extremes (6)
- National Security (5)
- Neutron Data Analysis and Visualization (2)
- Nuclear Science and Technology (17)
- Quantum Condensed Matter (2)
- Quantum information Science (3)
- Reactor Technology (1)
- Supercomputing (51)
- Transportation Systems (3)
Media Contacts
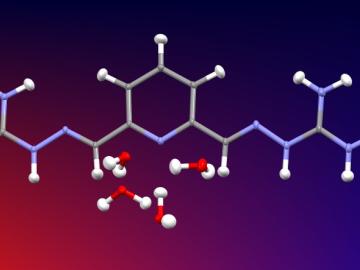
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate the effectiveness of a novel crystallization method to capture carbon dioxide directly from the air.
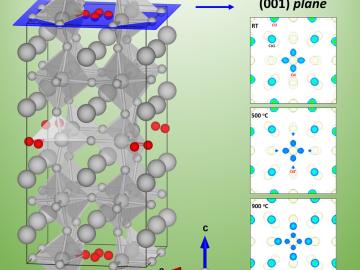
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.
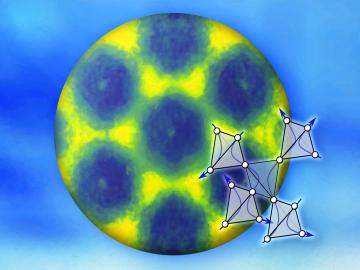
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate bizarre magnetic behavior, believed to be a possible quantum spin liquid rarely found in a three-dimensional material. QSLs are exotic states of matter where magnetism continues to fluctuate at low temperatures instead of “freezing” into aligned north and south poles as with traditional magnets.