
Filter News
Area of Research
- (-) Materials (23)
- Advanced Manufacturing (7)
- Biological Systems (1)
- Biology and Environment (1)
- Clean Energy (28)
- Climate and Environmental Systems (1)
- Computational Engineering (1)
- Computer Science (8)
- Fusion Energy (2)
- National Security (3)
- Neutron Science (6)
- Quantum information Science (3)
- Supercomputing (27)
- Transportation Systems (1)
News Type
News Topics
- (-) 3-D Printing/Advanced Manufacturing (5)
- (-) Computer Science (3)
- (-) Materials Science (16)
- (-) Molten Salt (1)
- (-) Sustainable Energy (5)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Bioenergy (2)
- Clean Water (2)
- Composites (1)
- Cybersecurity (1)
- Energy Storage (5)
- Environment (5)
- Fusion (2)
- Isotopes (1)
- Microscopy (4)
- Nanotechnology (5)
- Neutron Science (4)
- Nuclear Energy (5)
- Physics (1)
- Polymers (1)
- Quantum Science (2)
- Transportation (5)
Media Contacts

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.
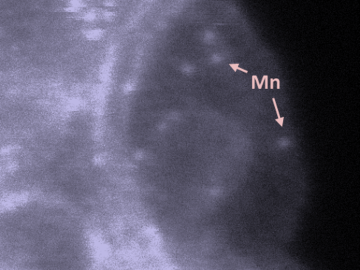
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.

Researchers have long sought electrically conductive materials for economical energy-storage devices. Two-dimensional (2D) ceramics called MXenes are contenders. Unlike most 2D ceramics, MXenes have inherently good conductivity because they are molecular sheets made from the carbides ...


