
Filter News
Area of Research
News Topics
- (-) Bioenergy (4)
- (-) Cybersecurity (1)
- (-) Isotopes (3)
- (-) Materials Science (9)
- (-) Microscopy (1)
- (-) Space Exploration (2)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (7)
- Artificial Intelligence (1)
- Big Data (1)
- Biomedical (6)
- Climate Change (1)
- Computer Science (7)
- Coronavirus (6)
- Decarbonization (1)
- Environment (2)
- Fusion (6)
- Machine Learning (1)
- Mathematics (1)
- Molten Salt (1)
- Nanotechnology (5)
- National Security (1)
- Neutron Science (28)
- Nuclear Energy (18)
- Physics (4)
- Polymers (1)
- Quantum Science (3)
- Security (1)
- Summit (5)
- Sustainable Energy (2)
- Transformational Challenge Reactor (3)
- Transportation (2)
Media Contacts
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.

Oak Ridge National Laboratory researchers have discovered a better way to separate actinium-227, a rare isotope essential for an FDA-approved cancer treatment.

Scientists at the Department of Energy Manufacturing Demonstration Facility at ORNL have their eyes on the prize: the Transformational Challenge Reactor, or TCR, a microreactor built using 3D printing and other new approaches that will be up and running by 2023.

Research by an international team led by Duke University and the Department of Energy’s Oak Ridge National Laboratory scientists could speed the way to safer rechargeable batteries for consumer electronics such as laptops and cellphones.

Biological membranes, such as the “walls” of most types of living cells, primarily consist of a double layer of lipids, or “lipid bilayer,” that forms the structure, and a variety of embedded and attached proteins with highly specialized functions, including proteins that rapidly and selectively transport ions and molecules in and out of the cell.
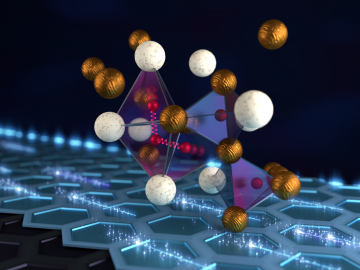
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.


