
Filter News
Area of Research
- Advanced Manufacturing (2)
- Biological Systems (1)
- Biology and Environment (3)
- Clean Energy (41)
- Climate and Environmental Systems (3)
- Computer Science (2)
- Energy Frontier Research Centers (1)
- Materials (29)
- National Security (2)
- Neutron Science (11)
- Nuclear Science and Technology (3)
- Quantum information Science (2)
- Supercomputing (23)
- Transportation Systems (1)
News Topics
- (-) Bioenergy (13)
- (-) Composites (6)
- (-) Cybersecurity (10)
- (-) Energy Storage (11)
- (-) Environment (26)
- (-) Frontier (2)
- (-) Molten Salt (5)
- (-) Nanotechnology (15)
- (-) Summit (11)
- (-) Transportation (21)
- 3-D Printing/Advanced Manufacturing (21)
- Advanced Reactors (8)
- Artificial Intelligence (14)
- Big Data (10)
- Biology (2)
- Biomedical (9)
- Biotechnology (1)
- Clean Water (6)
- Climate Change (1)
- Computer Science (55)
- Critical Materials (1)
- Exascale Computing (3)
- Fusion (10)
- Grid (8)
- Isotopes (7)
- Machine Learning (5)
- Materials Science (32)
- Mercury (3)
- Microscopy (11)
- Neutron Science (28)
- Nuclear Energy (27)
- Physics (17)
- Polymers (7)
- Quantum Science (13)
- Security (9)
- Space Exploration (6)
- Sustainable Energy (8)
Media Contacts
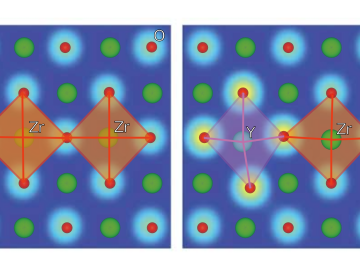
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
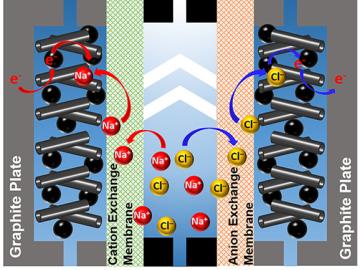
A team of scientists led by Oak Ridge National Laboratory used carbon nanotubes to improve a desalination process that attracts and removes ionic compounds such as salt from water using charged electrodes.

OAK RIDGE, Tenn., March 11, 2019—An international collaboration including scientists at the Department of Energy’s Oak Ridge National Laboratory solved a 50-year-old puzzle that explains why beta decays of atomic nuclei
Higher carbon dioxide levels caused 30 percent more wood growth in young forest stands across the temperate United States over a decade, according to an analysis led by Oak Ridge National Laboratory.

Oak Ridge National Laboratory’s latest Transportation Energy Data Book: Edition 37 reports that the number of vehicles nationwide is growing faster than the population, with sales more than 17 million since 2015, and the average household vehicle travels more than 11,000 miles per year.
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

Vera Bocharova at the Department of Energy’s Oak Ridge National Laboratory investigates the structure and dynamics of soft materials—polymer nanocomposites, polymer electrolytes and biological macromolecules—to advance materials and technologies for energy, medicine and other applications.

The use of lithium-ion batteries has surged in recent years, starting with electronics and expanding into many applications, including the growing electric and hybrid vehicle industry. But the technologies to optimize recycling of these batteries have not kept pace.
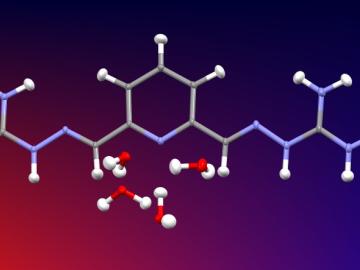
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate the effectiveness of a novel crystallization method to capture carbon dioxide directly from the air.


