Filter News
Area of Research
News Topics
- (-) Isotopes (2)
- (-) Nanotechnology (2)
- 3-D Printing/Advanced Manufacturing (3)
- Advanced Reactors (1)
- Artificial Intelligence (1)
- Bioenergy (2)
- Biomedical (1)
- Composites (1)
- Computer Science (7)
- Cybersecurity (1)
- Environment (1)
- Microscopy (1)
- Neutron Science (3)
- Nuclear Energy (2)
- Physics (3)
- Quantum Science (2)
- Security (3)
- Summit (2)
Media Contacts
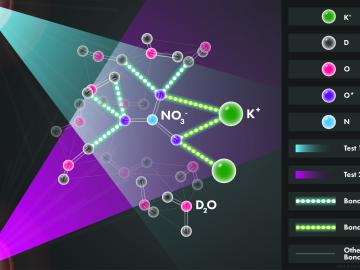
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

Scientists at the Department of Energy’s Oak Ridge National Laboratory induced a two-dimensional material to cannibalize itself for atomic “building blocks” from which stable structures formed. The findings, reported in Nature Communications, provide insights that ...

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.

A scientific team led by the Department of Energy’s Oak Ridge National Laboratory has found a new way to take the local temperature of a material from an area about a billionth of a meter wide, or approximately 100,000 times thinner than a human hair. This discove...




