
Filter News
Area of Research
News Topics
- (-) Energy Storage (29)
- (-) Transportation (30)
- 3-D Printing/Advanced Manufacturing (35)
- Advanced Reactors (10)
- Artificial Intelligence (38)
- Big Data (17)
- Bioenergy (31)
- Biology (29)
- Biomedical (12)
- Biotechnology (7)
- Buildings (14)
- Chemical Sciences (24)
- Clean Water (10)
- Climate Change (31)
- Composites (8)
- Computer Science (58)
- Coronavirus (4)
- Critical Materials (6)
- Cybersecurity (14)
- Decarbonization (30)
- Education (3)
- Emergency (1)
- Environment (62)
- Exascale Computing (17)
- Fossil Energy (2)
- Frontier (21)
- Fusion (14)
- Grid (21)
- High-Performance Computing (33)
- Hydropower (3)
- Irradiation (2)
- Isotopes (12)
- Machine Learning (20)
- Materials (59)
- Materials Science (36)
- Mathematics (2)
- Mercury (3)
- Microelectronics (2)
- Microscopy (12)
- Molten Salt (2)
- Nanotechnology (13)
- National Security (21)
- Net Zero (5)
- Neutron Science (50)
- Nuclear Energy (38)
- Partnerships (24)
- Physics (20)
- Polymers (6)
- Quantum Computing (12)
- Quantum Science (19)
- Renewable Energy (2)
- Security (5)
- Simulation (29)
- Software (1)
- Space Exploration (8)
- Summit (18)
- Sustainable Energy (25)
Media Contacts

Researchers at Oak Ridge National Laboratory proved that a certain class of ionic liquids, when mixed with commercially available oils, can make gears run more efficiently with less noise and better durability.

In Hong Wang’s world, nothing is beyond control. Before joining Oak Ridge National Laboratory as a senior distinguished researcher in transportation systems, he spent more than three decades studying the control of complex industrial systems in the United Kingdom.
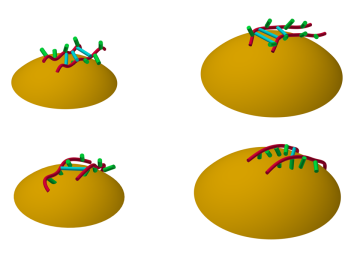
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

Galigekere is principal investigator for the breakthrough work in fast, wireless charging of electric vehicles being performed at the National Transportation Research Center at Oak Ridge National Laboratory.
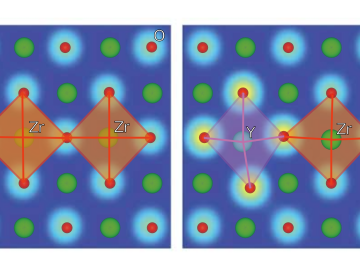
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

Oak Ridge National Laboratory’s latest Transportation Energy Data Book: Edition 37 reports that the number of vehicles nationwide is growing faster than the population, with sales more than 17 million since 2015, and the average household vehicle travels more than 11,000 miles per year.

The use of lithium-ion batteries has surged in recent years, starting with electronics and expanding into many applications, including the growing electric and hybrid vehicle industry. But the technologies to optimize recycling of these batteries have not kept pace.

Oak Ridge National Laboratory scientists have created open source software that scales up analysis of motor designs to run on the fastest computers available, including those accessible to outside users at the Oak Ridge Leadership Computing Facility.
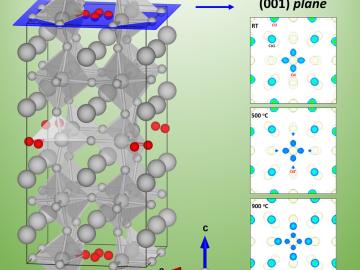
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.


