
Filter News
Area of Research
News Topics
- (-) Coronavirus (4)
- (-) Energy Storage (29)
- 3-D Printing/Advanced Manufacturing (35)
- Advanced Reactors (10)
- Artificial Intelligence (38)
- Big Data (17)
- Bioenergy (31)
- Biology (29)
- Biomedical (12)
- Biotechnology (7)
- Buildings (14)
- Chemical Sciences (24)
- Clean Water (10)
- Climate Change (31)
- Composites (8)
- Computer Science (58)
- Critical Materials (6)
- Cybersecurity (14)
- Decarbonization (30)
- Education (3)
- Emergency (1)
- Environment (62)
- Exascale Computing (17)
- Fossil Energy (2)
- Frontier (21)
- Fusion (14)
- Grid (21)
- High-Performance Computing (33)
- Hydropower (3)
- Irradiation (2)
- Isotopes (12)
- Machine Learning (20)
- Materials (59)
- Materials Science (36)
- Mathematics (2)
- Mercury (3)
- Microelectronics (2)
- Microscopy (12)
- Molten Salt (2)
- Nanotechnology (13)
- National Security (21)
- Net Zero (5)
- Neutron Science (50)
- Nuclear Energy (38)
- Partnerships (24)
- Physics (20)
- Polymers (6)
- Quantum Computing (12)
- Quantum Science (19)
- Renewable Energy (2)
- Security (5)
- Simulation (29)
- Software (1)
- Space Exploration (8)
- Summit (18)
- Sustainable Energy (25)
- Transportation (30)
Media Contacts

Andrew Ullman, Distinguished Staff Fellow at Oak Ridge National Laboratory, is using chemistry to devise a better battery
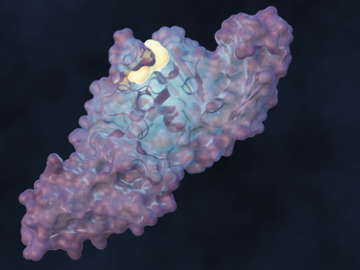
A team of scientists led by the Department of Energy’s Oak Ridge National Laboratory designed a molecule that disrupts the infection mechanism of the SARS-CoV-2 coronavirus and could be used to develop new treatments for COVID-19 and other viral diseases.
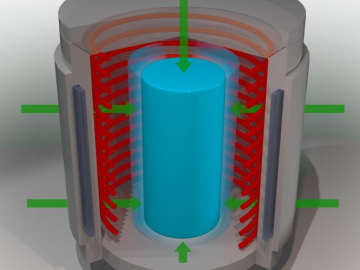
Following months of promising test results, battery researchers at ORNL are recommending that the solid-state battery industry focus on a technique known as isostatic pressing as it looks to commercialize next-generation batteries.

ORNL researchers Ben Ollis and Max Ferrari will be in Adjuntas to join the March 18 festivities but also to hammer out more technical details of their contribution to the project: making the microgrids even more reliable.

When aging vehicle batteries lack the juice to power your car anymore, they may still hold energy. Yet it’s tough to find new uses for lithium-ion batteries with different makers, ages and sizes. A solution is urgently needed because battery recycling options are scarce.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.
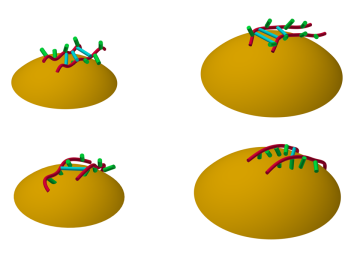
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
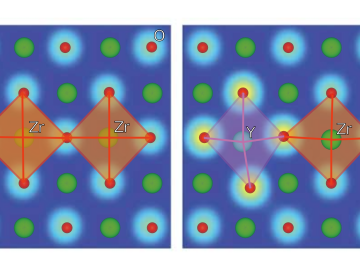
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

The use of lithium-ion batteries has surged in recent years, starting with electronics and expanding into many applications, including the growing electric and hybrid vehicle industry. But the technologies to optimize recycling of these batteries have not kept pace.


