
Filter News
Area of Research
- (-) National Security (6)
- (-) Neutron Science (20)
- Advanced Manufacturing (2)
- Biology and Environment (7)
- Clean Energy (24)
- Computational Engineering (1)
- Computer Science (2)
- Fusion and Fission (9)
- Fusion Energy (2)
- Isotopes (2)
- Materials (24)
- Materials for Computing (6)
- Quantum information Science (1)
- Supercomputing (24)
News Topics
- (-) Biomedical (3)
- (-) Computer Science (6)
- (-) Frontier (2)
- (-) Fusion (1)
- (-) Neutron Science (18)
- (-) Quantum Science (2)
- Artificial Intelligence (3)
- Biology (4)
- Chemical Sciences (2)
- Coronavirus (1)
- Cybersecurity (5)
- Decarbonization (1)
- Energy Storage (3)
- Environment (3)
- Exascale Computing (1)
- Grid (1)
- Machine Learning (2)
- Materials (5)
- Materials Science (5)
- Nanotechnology (1)
- National Security (7)
- Partnerships (1)
- Physics (4)
- Security (3)
- Space Exploration (1)
Media Contacts
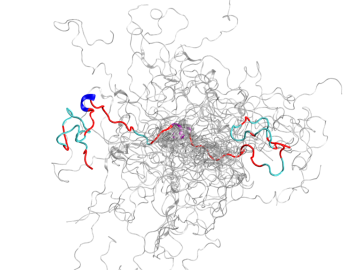
Using the Titan supercomputer and the Spallation Neutron Source at the Department of Energy’s Oak Ridge National Laboratory, scientists have created the most accurate 3D model yet of an intrinsically disordered protein, revealing the ensemble of its atomic-level structures.
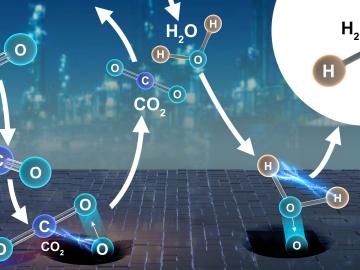
Collaborators at the Department of Energy’s Oak Ridge National Laboratory and U.S. universities used neutron scattering and other advanced characterization techniques to study how a prominent catalyst enables the “water-gas shift” reaction to purify and generate hydrogen at industrial scale.

An ORNL-led team's observation of certain crystalline ice phases challenges accepted theories about super-cooled water and non-crystalline ice. Their findings, reported in the journal Nature, will also lead to better understanding of ice and its various phases found on other planets, moons and elsewhere in space.

OAK RIDGE, Tenn., May 14, 2019—Advanced Research Systems, Inc., has licensed a technology designed to automatically refill liquid helium used in laboratory equipment for low-temperature scientific experiments, which will reduce downtime, recover more helium and increase overall efficiency.
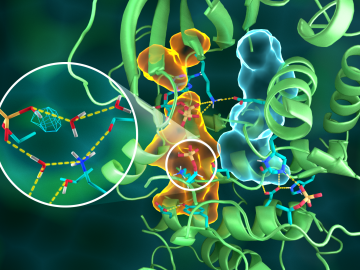
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.


