
Filter News
Area of Research
- (-) Clean Energy (52)
- (-) National Security (4)
- (-) Neutron Science (10)
- Advanced Manufacturing (3)
- Biology and Environment (5)
- Computational Engineering (1)
- Computer Science (7)
- Energy Sciences (1)
- Fusion Energy (1)
- Materials (26)
- Materials for Computing (3)
- Nuclear Science and Technology (1)
- Quantum information Science (6)
- Supercomputing (22)
News Topics
- (-) Clean Water (6)
- (-) Composites (3)
- (-) Energy Storage (22)
- (-) Grid (9)
- (-) Machine Learning (8)
- (-) Quantum Science (5)
- (-) Sustainable Energy (28)
- 3-D Printing/Advanced Manufacturing (30)
- Advanced Reactors (3)
- Artificial Intelligence (7)
- Big Data (6)
- Bioenergy (18)
- Biology (1)
- Biomedical (10)
- Biotechnology (2)
- Chemical Sciences (2)
- Climate Change (7)
- Computer Science (26)
- Coronavirus (13)
- Cybersecurity (6)
- Decarbonization (1)
- Environment (33)
- Exascale Computing (1)
- High-Performance Computing (1)
- Isotopes (1)
- Materials (2)
- Materials Science (23)
- Mathematics (2)
- Mercury (1)
- Microscopy (5)
- Molten Salt (1)
- Nanotechnology (11)
- National Security (2)
- Neutron Science (43)
- Nuclear Energy (7)
- Physics (5)
- Polymers (5)
- Security (4)
- Space Exploration (1)
- Summit (7)
- Transformational Challenge Reactor (2)
- Transportation (24)
Media Contacts
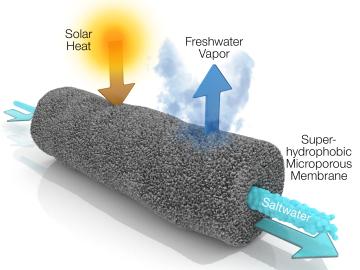
A new method developed at Oak Ridge National Laboratory improves the energy efficiency of a desalination process known as solar-thermal evaporation.
A team of scientists led by Oak Ridge National Laboratory have discovered the specific gene that controls an important symbiotic relationship between plants and soil fungi, and successfully facilitated the symbiosis in a plant that
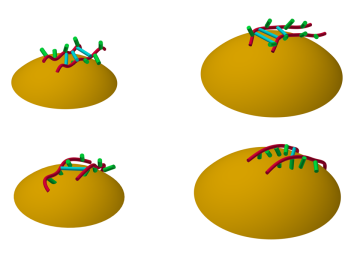
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.
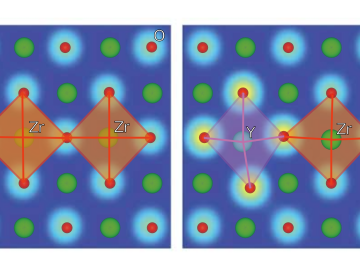
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

Scientists at Oak Ridge National Laboratory have developed a low-cost, printed, flexible sensor that can wrap around power cables to precisely monitor electrical loads from household appliances to support grid operations.
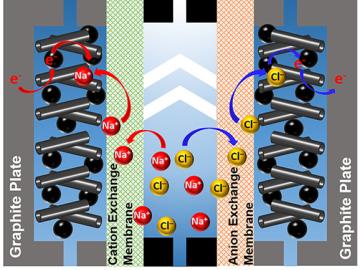
A team of scientists led by Oak Ridge National Laboratory used carbon nanotubes to improve a desalination process that attracts and removes ionic compounds such as salt from water using charged electrodes.

The use of lithium-ion batteries has surged in recent years, starting with electronics and expanding into many applications, including the growing electric and hybrid vehicle industry. But the technologies to optimize recycling of these batteries have not kept pace.
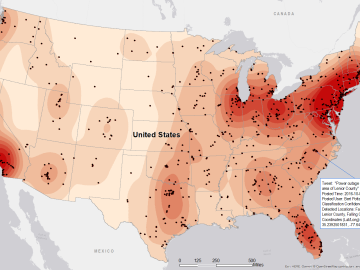
Gleaning valuable data from social platforms such as Twitter—particularly to map out critical location information during emergencies— has become more effective and efficient thanks to Oak Ridge National Laboratory.
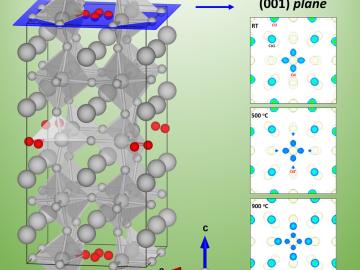
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.


