
Filter News
Area of Research
- (-) National Security (9)
- (-) Neutron Science (19)
- (-) Nuclear Systems Modeling, Simulation and Validation (1)
- Advanced Manufacturing (5)
- Biology and Environment (7)
- Clean Energy (50)
- Computational Engineering (1)
- Computer Science (6)
- Energy Sciences (1)
- Fusion and Fission (4)
- Fusion Energy (8)
- Isotopes (2)
- Materials (59)
- Materials for Computing (5)
- Nuclear Science and Technology (27)
- Supercomputing (40)
- Transportation Systems (1)
News Topics
- (-) Artificial Intelligence (3)
- (-) Energy Storage (4)
- (-) Materials Science (11)
- (-) Nuclear Energy (5)
- (-) Summit (6)
- (-) Transportation (1)
- 3-D Printing/Advanced Manufacturing (2)
- Advanced Reactors (2)
- Big Data (4)
- Bioenergy (4)
- Biomedical (7)
- Clean Water (1)
- Climate Change (1)
- Computer Science (14)
- Coronavirus (6)
- Cybersecurity (4)
- Environment (7)
- Grid (2)
- Machine Learning (4)
- Mathematics (1)
- Microscopy (2)
- Nanotechnology (6)
- National Security (2)
- Neutron Science (43)
- Physics (4)
- Polymers (1)
- Quantum Science (4)
- Security (4)
- Sustainable Energy (2)
Media Contacts

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.

IDEMIA Identity & Security USA has licensed an advanced optical array developed at Oak Ridge National Laboratory. The portable technology can be used to help identify individuals in challenging outdoor conditions.
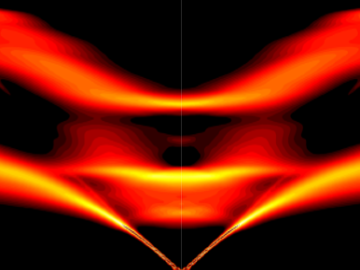
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials

Researchers at the Department of Energy’s Oak Ridge National Laboratory, Pacific Northwest National Laboratory and Washington State University teamed up to investigate the complex dynamics of low-water liquids that challenge nuclear waste processing at federal cleanup sites.
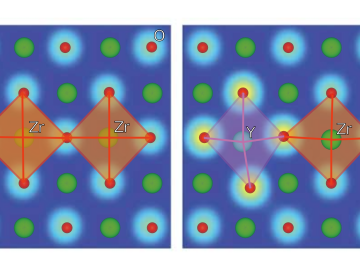
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

Scientists at the Department of Energy’s Oak Ridge National Laboratory are working to understand both the complex nature of uranium and the various oxide forms it can take during processing steps that might occur throughout the nuclear fuel cycle.
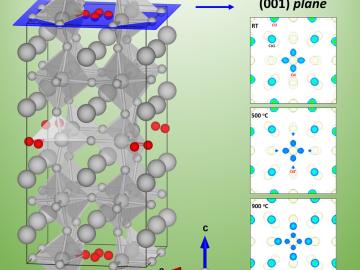
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.


