
Filter News
Area of Research
News Topics
- (-) Artificial Intelligence (2)
- (-) Computer Science (7)
- (-) Energy Storage (1)
- (-) Machine Learning (2)
- (-) Microscopy (2)
- (-) Nuclear Energy (2)
- (-) Physics (3)
- (-) Quantum Science (4)
- (-) Sustainable Energy (3)
- 3-D Printing/Advanced Manufacturing (5)
- Bioenergy (3)
- Biology (1)
- Biomedical (3)
- Chemical Sciences (1)
- Climate Change (1)
- Coronavirus (5)
- Decarbonization (1)
- Environment (3)
- Grid (1)
- High-Performance Computing (1)
- Isotopes (2)
- Materials (2)
- Materials Science (6)
- Molten Salt (1)
- Nanotechnology (6)
- National Security (1)
- Neutron Science (13)
- Summit (5)
- Transformational Challenge Reactor (1)
- Transportation (2)
Media Contacts

Scientists at ORNL used neutron scattering and supercomputing to better understand how an organic solvent and water work together to break down plant biomass, creating a pathway to significantly improve the production of renewable

Scientists from the Department of Energy’s Oak Ridge National Laboratory and a dozen other international research institutions have produced the most elaborate set of projections to date that illustrates possible futures for major monsoon regions.
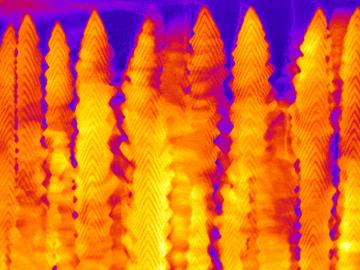
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.

For the second year in a row, a team from the Department of Energy’s Oak Ridge and Los Alamos national laboratories led a demonstration hosted by EPB, a community-based utility and telecommunications company serving Chattanooga, Tennessee.
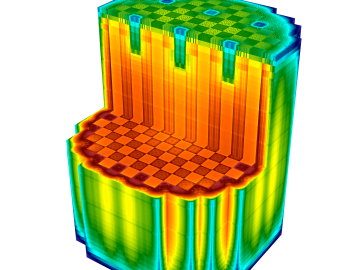
OAK RIDGE, Tenn., Feb. 19, 2020 — The U.S. Department of Energy’s Oak Ridge National Laboratory and the Tennessee Valley Authority have signed a memorandum of understanding to evaluate a new generation of flexible, cost-effective advanced nuclear reactors.
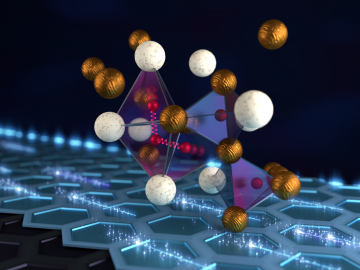
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.


