
Filter News
Area of Research
- (-) Materials (53)
- (-) Nuclear Science and Technology (4)
- Advanced Manufacturing (2)
- Biology and Environment (16)
- Building Technologies (1)
- Clean Energy (37)
- Climate and Environmental Systems (1)
- Computational Engineering (3)
- Computer Science (13)
- Energy Frontier Research Centers (1)
- Fusion and Fission (2)
- Fusion Energy (2)
- Isotopes (10)
- Materials for Computing (12)
- Mathematics (1)
- National Security (13)
- Neutron Science (14)
- Quantum information Science (4)
- Supercomputing (57)
News Type
News Topics
- (-) Artificial Intelligence (4)
- (-) Computer Science (10)
- (-) Isotopes (9)
- (-) Machine Learning (2)
- (-) Nanotechnology (29)
- (-) Polymers (12)
- 3-D Printing/Advanced Manufacturing (21)
- Advanced Reactors (8)
- Bioenergy (10)
- Biology (4)
- Biomedical (6)
- Buildings (3)
- Chemical Sciences (24)
- Clean Water (1)
- Climate Change (5)
- Composites (7)
- Coronavirus (3)
- Critical Materials (13)
- Cybersecurity (3)
- Decarbonization (6)
- Energy Storage (26)
- Environment (8)
- Exascale Computing (1)
- Frontier (2)
- Fusion (6)
- Grid (2)
- High-Performance Computing (2)
- ITER (1)
- Materials (50)
- Materials Science (55)
- Microscopy (18)
- Molten Salt (6)
- National Security (3)
- Net Zero (1)
- Neutron Science (25)
- Nuclear Energy (22)
- Partnerships (8)
- Physics (17)
- Quantum Computing (2)
- Quantum Science (11)
- Renewable Energy (1)
- Security (1)
- Space Exploration (5)
- Summit (1)
- Sustainable Energy (11)
- Transformational Challenge Reactor (2)
- Transportation (10)
Media Contacts

An all-in-one experimental platform developed at Oak Ridge National Laboratory’s Center for Nanophase Materials Sciences accelerates research on promising materials for future technologies.

Real-time measurements captured by researchers at ORNL provide missing insight into chemical separations to recover cobalt, a critical raw material used to make batteries and magnets for modern technologies.

After its long journey to Mars beginning this summer, NASA’s Perseverance rover will be powered across the planet’s surface in part by plutonium produced at the Department of Energy’s Oak Ridge National Laboratory.

Five researchers at the Department of Energy’s Oak Ridge National Laboratory have been named ORNL Corporate Fellows in recognition of significant career accomplishments and continued leadership in their scientific fields.
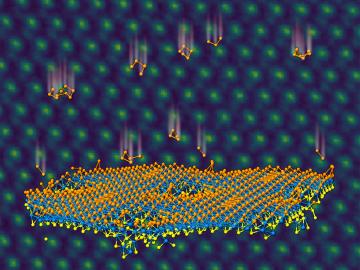
An ORNL team used a simple process to implant atoms precisely into the top layers of ultra-thin crystals, yielding two-sided structures with different chemical compositions.
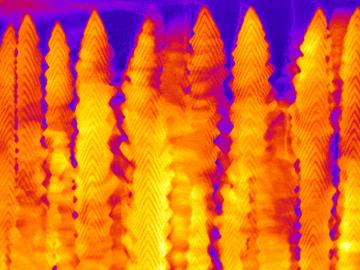
A team led by the Department of Energy’s Oak Ridge National Laboratory synthesized a tiny structure with high surface area and discovered how its unique architecture drives ions across interfaces to transport energy or information.

Oak Ridge National Laboratory researchers have discovered a better way to separate actinium-227, a rare isotope essential for an FDA-approved cancer treatment.

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.

OAK RIDGE, Tenn., Feb. 27, 2020 — Researchers at Oak Ridge National Laboratory and the University of Tennessee achieved a rare look at the inner workings of polymer self-assembly at an oil-water interface to advance materials for neuromorphic computing and bio-inspired technologies.
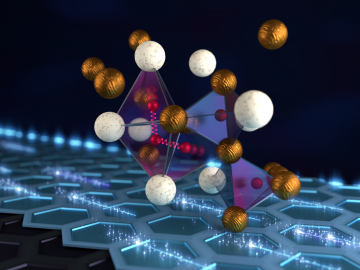
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.


