
Filter News
Area of Research
- (-) Materials (45)
- Advanced Manufacturing (1)
- Biology and Environment (41)
- Biology and Soft Matter (1)
- Clean Energy (80)
- Climate and Environmental Systems (1)
- Computer Science (2)
- Fusion and Fission (5)
- Isotope Development and Production (1)
- Isotopes (23)
- Materials for Computing (8)
- National Security (24)
- Neutron Science (10)
- Nuclear Science and Technology (6)
- Quantum information Science (1)
- Supercomputing (64)
News Type
News Topics
- (-) Buildings (3)
- (-) Climate Change (5)
- (-) Cybersecurity (4)
- (-) Energy Storage (25)
- (-) Frontier (2)
- (-) Isotopes (11)
- (-) Molten Salt (2)
- (-) Quantum Computing (2)
- (-) Space Exploration (1)
- 3-D Printing/Advanced Manufacturing (17)
- Advanced Reactors (2)
- Artificial Intelligence (8)
- Big Data (2)
- Bioenergy (10)
- Biology (4)
- Biomedical (5)
- Chemical Sciences (27)
- Clean Water (2)
- Composites (5)
- Computer Science (16)
- Coronavirus (3)
- Critical Materials (8)
- Decarbonization (5)
- Environment (13)
- Exascale Computing (2)
- Fusion (4)
- Grid (4)
- High-Performance Computing (3)
- ITER (1)
- Machine Learning (4)
- Materials (57)
- Materials Science (52)
- Mathematics (1)
- Microscopy (18)
- Nanotechnology (29)
- National Security (3)
- Net Zero (1)
- Neutron Science (27)
- Nuclear Energy (11)
- Partnerships (11)
- Physics (25)
- Polymers (10)
- Quantum Science (10)
- Renewable Energy (1)
- Security (2)
- Summit (2)
- Sustainable Energy (9)
- Transformational Challenge Reactor (3)
- Transportation (8)
Media Contacts
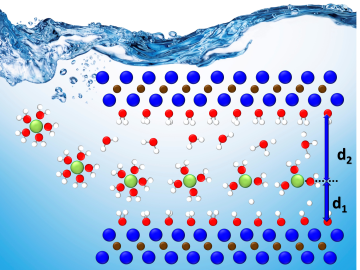
A team led by Oak Ridge National Laboratory developed a novel, integrated approach to track energy-transporting ions within an ultra-thin material, which could unlock its energy storage potential leading toward faster charging, longer-lasting devices.
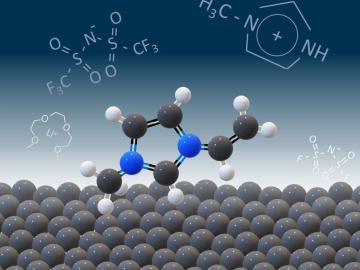
Scientists seeking ways to improve a battery’s ability to hold a charge longer, using advanced materials that are safe, stable and efficient, have determined that the materials themselves are only part of the solution.
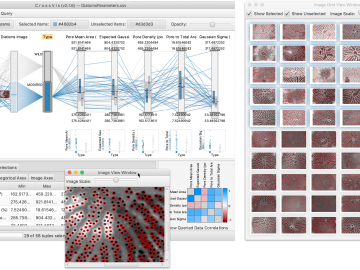
From materials science and earth system modeling to quantum information science and cybersecurity, experts in many fields run simulations and conduct experiments to collect the abundance of data necessary for scientific progress.

Energy storage startup SPARKZ Inc. has exclusively licensed five battery technologies from the Department of Energy’s Oak Ridge National Laboratory designed to eliminate cobalt metal in lithium-ion batteries. The advancement is aimed at accelerating the production of electric vehicles and energy storage solutions for the power grid.

The formation of lithium dendrites is still a mystery, but materials engineers study the conditions that enable dendrites and how to stop them.

Researchers at the Department of Energy’s Oak Ridge National Laboratory have received five 2019 R&D 100 Awards, increasing the lab’s total to 221 since the award’s inception in 1963.
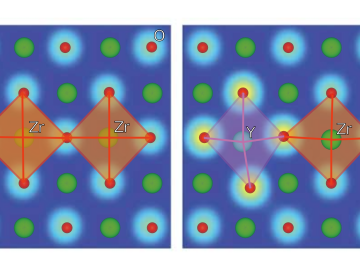
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
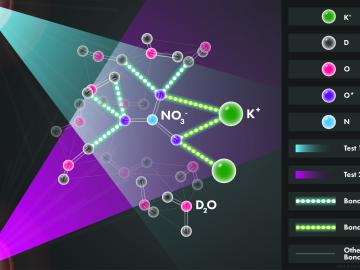
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come


