
Filter News
Area of Research
- Advanced Manufacturing (2)
- Biology and Environment (17)
- Clean Energy (45)
- Computational Engineering (1)
- Computer Science (2)
- Fusion and Fission (7)
- Fusion Energy (1)
- Isotope Development and Production (1)
- Isotopes (7)
- Materials (57)
- Materials Characterization (1)
- Materials for Computing (8)
- Materials Under Extremes (1)
- National Security (13)
- Neutron Science (44)
- Nuclear Science and Technology (7)
- Quantum information Science (1)
- Supercomputing (24)
News Type
News Topics
- (-) Advanced Reactors (10)
- (-) Biomedical (17)
- (-) Clean Water (2)
- (-) Cybersecurity (17)
- (-) Isotopes (18)
- (-) Materials Science (50)
- (-) Neutron Science (49)
- (-) Sustainable Energy (30)
- 3-D Printing/Advanced Manufacturing (43)
- Artificial Intelligence (31)
- Big Data (7)
- Bioenergy (23)
- Biology (21)
- Biotechnology (7)
- Buildings (13)
- Chemical Sciences (28)
- Climate Change (22)
- Composites (10)
- Computer Science (58)
- Coronavirus (17)
- Critical Materials (11)
- Decarbonization (19)
- Education (3)
- Element Discovery (1)
- Energy Storage (42)
- Environment (35)
- Exascale Computing (9)
- Fossil Energy (1)
- Frontier (15)
- Fusion (14)
- Grid (15)
- High-Performance Computing (28)
- ITER (2)
- Machine Learning (13)
- Materials (57)
- Mercury (2)
- Microelectronics (1)
- Microscopy (16)
- Molten Salt (2)
- Nanotechnology (26)
- National Security (18)
- Net Zero (3)
- Nuclear Energy (26)
- Partnerships (29)
- Physics (24)
- Polymers (12)
- Quantum Computing (9)
- Quantum Science (26)
- Renewable Energy (1)
- Security (11)
- Simulation (8)
- Space Exploration (3)
- Statistics (1)
- Summit (21)
- Transformational Challenge Reactor (4)
- Transportation (25)
Media Contacts
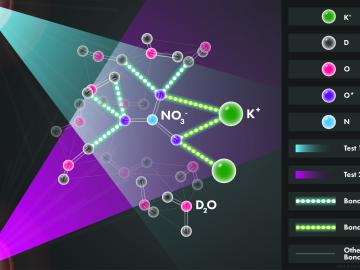
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

Qrypt, Inc., has exclusively licensed a novel cyber security technology from the Department of Energy’s Oak Ridge National Laboratory, promising a stronger defense against cyberattacks including those posed by quantum computing.

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.

After more than a year of operation at the Department of Energy’s (DOE’s) Oak Ridge National Laboratory (ORNL), the COHERENT experiment, using the world’s smallest neutrino detector, has found a big fingerprint of the elusive, electrically neutral particles that interact only weakly with matter.

Researchers used neutrons to probe a running engine at ORNL’s Spallation Neutron Source

Virginia-based Lenvio Inc. has exclusively licensed a cyber security technology from the Department of Energy’s Oak Ridge National Laboratory that can quickly detect malicious behavior in software not previously identified as a threat.

With the production of 50 grams of plutonium-238, researchers at the Department of Energy’s Oak Ridge National Laboratory have restored a U.S. capability dormant for nearly 30 years and set the course to provide power for NASA and other missions.
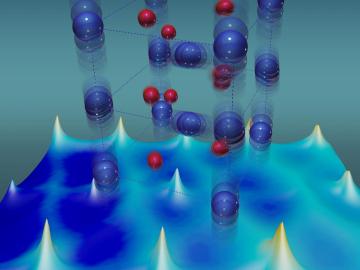
For more than 50 years, scientists have debated what turns particular oxide insulators, in which electrons barely move, into metals, in which electrons flow freely.


