
Filter News
Area of Research
- (-) Fusion and Fission (6)
- (-) Materials (67)
- Advanced Manufacturing (1)
- Biology and Environment (36)
- Clean Energy (87)
- Climate and Environmental Systems (1)
- Computational Biology (1)
- Computational Engineering (2)
- Computer Science (2)
- Electricity and Smart Grid (1)
- Energy Frontier Research Centers (1)
- Fusion Energy (7)
- Isotopes (3)
- Materials for Computing (10)
- Mathematics (1)
- National Security (9)
- Neutron Science (64)
- Nuclear Science and Technology (12)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Quantum information Science (1)
- Sensors and Controls (1)
- Supercomputing (29)
- Transportation Systems (2)
News Type
News Topics
- (-) Advanced Reactors (6)
- (-) Biomedical (5)
- (-) Climate Change (5)
- (-) Grid (3)
- (-) Molten Salt (3)
- (-) Nanotechnology (29)
- (-) Neutron Science (23)
- (-) Transportation (11)
- 3-D Printing/Advanced Manufacturing (19)
- Artificial Intelligence (4)
- Bioenergy (9)
- Biology (5)
- Buildings (3)
- Chemical Sciences (25)
- Clean Water (1)
- Composites (7)
- Computer Science (10)
- Coronavirus (3)
- Critical Materials (13)
- Cybersecurity (3)
- Decarbonization (6)
- Energy Storage (26)
- Environment (8)
- Exascale Computing (1)
- Fossil Energy (1)
- Frontier (2)
- Fusion (12)
- High-Performance Computing (2)
- Isotopes (7)
- ITER (4)
- Machine Learning (2)
- Materials (51)
- Materials Science (56)
- Microscopy (18)
- National Security (3)
- Net Zero (1)
- Nuclear Energy (14)
- Partnerships (10)
- Physics (16)
- Polymers (12)
- Quantum Computing (2)
- Quantum Science (11)
- Renewable Energy (1)
- Security (2)
- Simulation (1)
- Space Exploration (2)
- Summit (1)
- Sustainable Energy (11)
- Transformational Challenge Reactor (1)
Media Contacts

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.
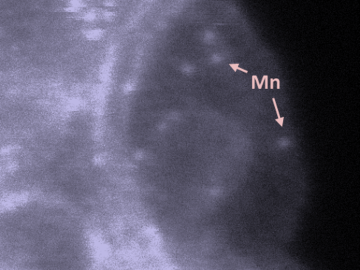
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.
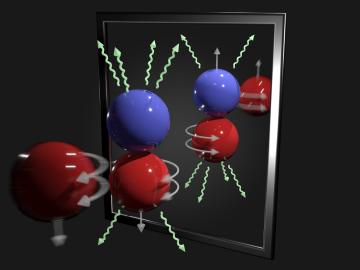
A team of scientists has for the first time measured the elusive weak interaction between protons and neutrons in the nucleus of an atom. They had chosen the simplest nucleus consisting of one neutron and one proton for the study.
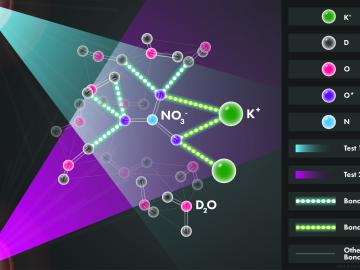
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come

An Oak Ridge National Laboratory-led team used a scanning transmission electron microscope to selectively position single atoms below a crystal’s surface for the first time.

Scientists at the Department of Energy’s Oak Ridge National Laboratory induced a two-dimensional material to cannibalize itself for atomic “building blocks” from which stable structures formed. The findings, reported in Nature Communications, provide insights that ...
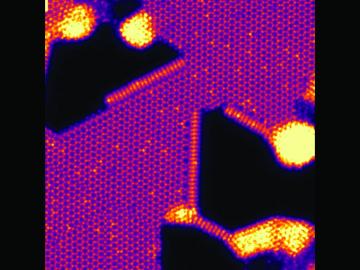
An Oak Ridge National Laboratory–led team has learned how to engineer tiny pores embellished with distinct edge structures inside atomically-thin two-dimensional, or 2D, crystals. The 2D crystals are envisioned as stackable building blocks for ultrathin electronics and other advance...

The Department of Energy’s Oak Ridge National Laboratory is now producing actinium-227 (Ac-227) to meet projected demand for a highly effective cancer drug through a 10-year contract between the U.S. DOE Isotope Program and Bayer.

A scientific team led by the Department of Energy’s Oak Ridge National Laboratory has found a new way to take the local temperature of a material from an area about a billionth of a meter wide, or approximately 100,000 times thinner than a human hair. This discove...


