
Filter News
Area of Research
- (-) Materials (18)
- Biology and Environment (18)
- Clean Energy (45)
- Climate and Environmental Systems (3)
- Computational Biology (1)
- Computational Engineering (2)
- Computer Science (8)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Fusion and Fission (1)
- Isotopes (1)
- Materials for Computing (4)
- Mathematics (1)
- National Security (3)
- Neutron Science (7)
- Quantum information Science (3)
- Sensors and Controls (1)
- Supercomputing (11)
News Topics
- (-) Energy Storage (7)
- (-) Environment (1)
- (-) Nanotechnology (8)
- (-) Physics (2)
- (-) Quantum Science (1)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Bioenergy (1)
- Biomedical (2)
- Buildings (1)
- Chemical Sciences (4)
- Clean Water (1)
- Composites (4)
- Computer Science (1)
- Coronavirus (1)
- Critical Materials (5)
- Decarbonization (1)
- Fusion (2)
- Isotopes (2)
- Materials (12)
- Materials Science (19)
- Microscopy (6)
- Molten Salt (1)
- Neutron Science (4)
- Nuclear Energy (3)
- Polymers (6)
- Quantum Computing (1)
- Space Exploration (1)
- Sustainable Energy (3)
- Transportation (6)
Media Contacts
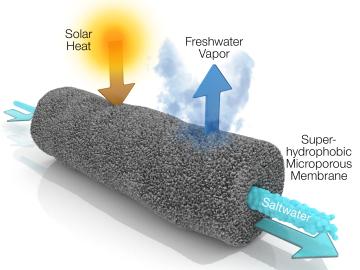
A new method developed at Oak Ridge National Laboratory improves the energy efficiency of a desalination process known as solar-thermal evaporation.
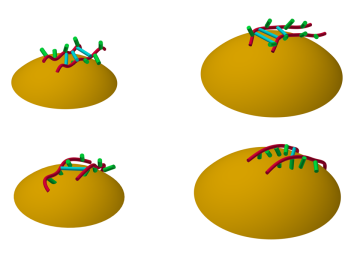
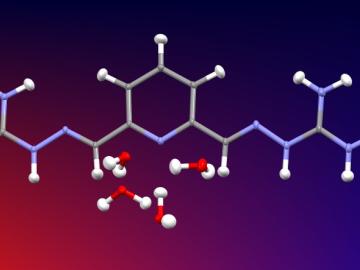
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.
Scientists have discovered a way to alter heat transport in thermoelectric materials, a finding that may ultimately improve energy efficiency as the materials
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate the effectiveness of a novel crystallization method to capture carbon dioxide directly from the air.
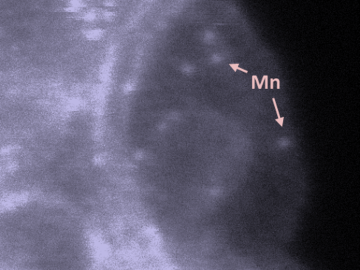
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.
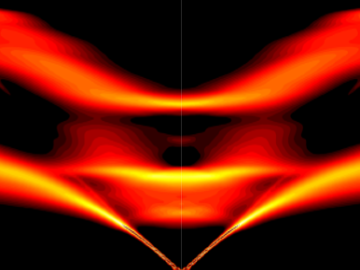
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.

An Oak Ridge National Laboratory-led team used a scanning transmission electron microscope to selectively position single atoms below a crystal’s surface for the first time.
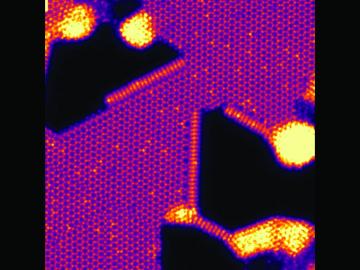
An Oak Ridge National Laboratory–led team has learned how to engineer tiny pores embellished with distinct edge structures inside atomically-thin two-dimensional, or 2D, crystals. The 2D crystals are envisioned as stackable building blocks for ultrathin electronics and other advance...


