
Filter News
Area of Research
- (-) Climate and Environmental Systems (5)
- (-) Computational Engineering (3)
- (-) Neutron Science (108)
- Advanced Manufacturing (5)
- Biological Systems (2)
- Biology and Environment (136)
- Biology and Soft Matter (1)
- Clean Energy (153)
- Computational Biology (2)
- Computer Science (10)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Functional Materials for Energy (2)
- Fusion and Fission (26)
- Fusion Energy (13)
- Isotopes (2)
- Materials (94)
- Materials for Computing (10)
- Mathematics (1)
- National Security (31)
- Nuclear Science and Technology (14)
- Quantum information Science (1)
- Supercomputing (102)
News Topics
- (-) Artificial Intelligence (7)
- (-) Bioenergy (6)
- (-) Energy Storage (6)
- (-) Environment (14)
- (-) Fusion (1)
- (-) Machine Learning (4)
- (-) Mercury (1)
- (-) Neutron Science (99)
- (-) Summit (7)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Big Data (3)
- Biology (6)
- Biomedical (12)
- Biotechnology (1)
- Chemical Sciences (2)
- Clean Water (3)
- Climate Change (4)
- Composites (1)
- Computer Science (17)
- Coronavirus (8)
- Cybersecurity (1)
- Decarbonization (2)
- Fossil Energy (1)
- Frontier (1)
- High-Performance Computing (3)
- Materials (14)
- Materials Science (23)
- Mathematics (2)
- Microscopy (3)
- Nanotechnology (10)
- National Security (2)
- Nuclear Energy (3)
- Physics (9)
- Polymers (1)
- Quantum Computing (1)
- Quantum Science (7)
- Security (2)
- Space Exploration (3)
- Sustainable Energy (2)
- Transportation (5)
Media Contacts
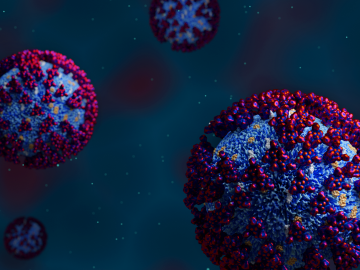
In the race to identify solutions to the COVID-19 pandemic, researchers at the Department of Energy’s Oak Ridge National Laboratory are joining the fight by applying expertise in computational science, advanced manufacturing, data science and neutron science.
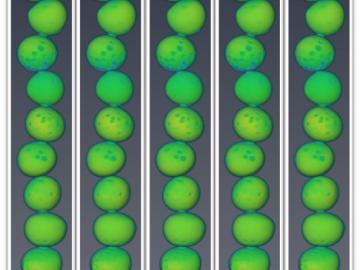
Oak Ridge National Laboratory researchers working on neutron imaging capabilities for nuclear materials have developed a process for seeing the inside of uranium particles – without cutting them open.

Biological membranes, such as the “walls” of most types of living cells, primarily consist of a double layer of lipids, or “lipid bilayer,” that forms the structure, and a variety of embedded and attached proteins with highly specialized functions, including proteins that rapidly and selectively transport ions and molecules in and out of the cell.
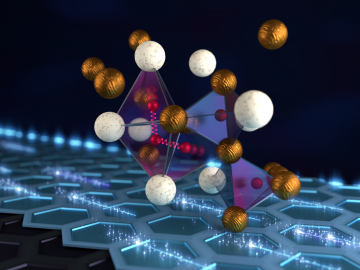
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.
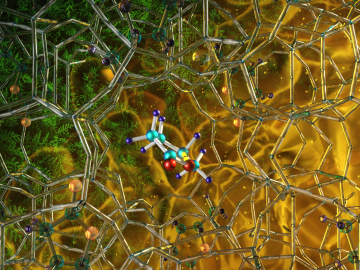
Illustration of the optimized zeolite catalyst, or NbAlS-1, which enables a highly efficient chemical reaction to create butene, a renewable source of energy, without expending high amounts of energy for the conversion. Credit: Jill Hemman, Oak Ridge National Laboratory/U.S. Dept. of Energy

ORNL computer scientist Catherine Schuman returned to her alma mater, Harriman High School, to lead Hour of Code activities and talk to students about her job as a researcher.
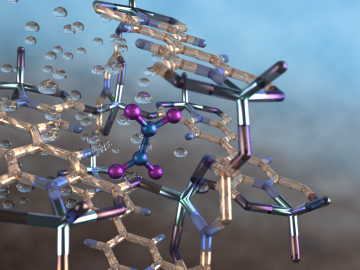
An international team of scientists, led by the University of Manchester, has developed a metal-organic framework, or MOF, material

Scientists at the U.S. Department of Energy’s Brookhaven National Laboratory have new experimental evidence and a predictive theory that solves a long-standing materials science mystery: why certain crystalline materials shrink when heated.

In the vast frozen whiteness of the central Arctic, the Polarstern, a German research vessel, has settled into the ice for a yearlong float.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.


