
Filter News
Area of Research
- (-) Energy Frontier Research Centers (1)
- (-) Materials for Computing (15)
- Advanced Manufacturing (5)
- Biology and Environment (79)
- Biology and Soft Matter (1)
- Building Technologies (2)
- Clean Energy (126)
- Climate and Environmental Systems (2)
- Computational Engineering (1)
- Computer Science (4)
- Electricity and Smart Grid (3)
- Energy Sciences (1)
- Functional Materials for Energy (1)
- Fusion and Fission (8)
- Fusion Energy (2)
- Isotope Development and Production (1)
- Isotopes (5)
- Materials (70)
- Mathematics (1)
- National Security (28)
- Neutron Science (18)
- Nuclear Science and Technology (7)
- Quantum information Science (4)
- Sensors and Controls (1)
- Supercomputing (70)
News Topics
- (-) Climate Change (1)
- (-) Microscopy (4)
- (-) Nanotechnology (8)
- (-) Space Exploration (1)
- (-) Sustainable Energy (5)
- 3-D Printing/Advanced Manufacturing (4)
- Bioenergy (1)
- Biology (1)
- Biomedical (2)
- Chemical Sciences (4)
- Composites (1)
- Computer Science (7)
- Coronavirus (3)
- Decarbonization (1)
- Energy Storage (4)
- Environment (1)
- Isotopes (1)
- Materials (10)
- Materials Science (15)
- National Security (1)
- Neutron Science (5)
- Polymers (6)
- Quantum Computing (1)
- Quantum Science (3)
- Security (1)
- Simulation (1)
- Summit (1)
- Transportation (5)
Media Contacts

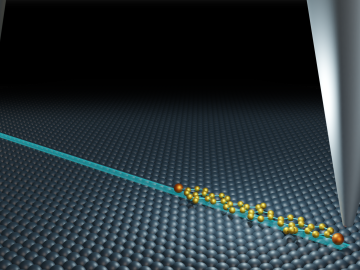
Oak Ridge National Laboratory’s Center for Nanophase Materials Sciences contributed to a groundbreaking experiment published in Science that tracks the real-time transport of individual molecules.
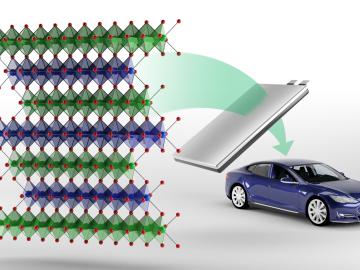
Oak Ridge National Laboratory researchers have developed a new family of cathodes with the potential to replace the costly cobalt-based cathodes typically found in today’s lithium-ion batteries that power electric vehicles and consumer electronics.
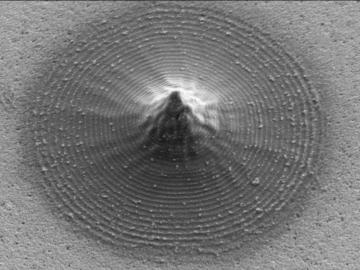
Scientists at Oak Ridge National Laboratory and the University of Tennessee designed and demonstrated a method to make carbon-based materials that can be used as electrodes compatible with a specific semiconductor circuitry.

Four research teams from the Department of Energy’s Oak Ridge National Laboratory and their technologies have received 2020 R&D 100 Awards.

Scientists at the Department of Energy’s Oak Ridge National Laboratory induced a two-dimensional material to cannibalize itself for atomic “building blocks” from which stable structures formed. The findings, reported in Nature Communications, provide insights that ...


