
Filter News
Area of Research
- (-) Materials (85)
- (-) Nuclear Science and Technology (10)
- Advanced Manufacturing (2)
- Biological Systems (1)
- Biology and Environment (112)
- Biology and Soft Matter (1)
- Clean Energy (148)
- Climate and Environmental Systems (5)
- Computational Biology (2)
- Computational Engineering (3)
- Computer Science (6)
- Electricity and Smart Grid (1)
- Functional Materials for Energy (1)
- Fusion and Fission (7)
- Isotope Development and Production (1)
- Isotopes (28)
- Materials for Computing (17)
- Mathematics (1)
- National Security (36)
- Neutron Science (33)
- Quantum information Science (4)
- Sensors and Controls (1)
- Supercomputing (74)
- Transportation Systems (2)
News Topics
- (-) Big Data (2)
- (-) Biomedical (8)
- (-) Cybersecurity (5)
- (-) Environment (16)
- (-) Isotopes (16)
- (-) Microscopy (27)
- (-) Polymers (17)
- (-) Security (2)
- (-) Space Exploration (7)
- (-) Transportation (14)
- 3-D Printing/Advanced Manufacturing (26)
- Advanced Reactors (14)
- Artificial Intelligence (9)
- Bioenergy (12)
- Biology (4)
- Buildings (5)
- Chemical Sciences (32)
- Clean Water (3)
- Climate Change (5)
- Composites (9)
- Computer Science (19)
- Coronavirus (5)
- Critical Materials (13)
- Decarbonization (8)
- Energy Storage (34)
- Exascale Computing (2)
- Frontier (3)
- Fusion (15)
- Grid (5)
- High-Performance Computing (4)
- Irradiation (1)
- ITER (1)
- Machine Learning (5)
- Materials (73)
- Materials Science (79)
- Mathematics (1)
- Molten Salt (7)
- Nanotechnology (39)
- National Security (3)
- Net Zero (1)
- Neutron Science (38)
- Nuclear Energy (49)
- Partnerships (11)
- Physics (31)
- Quantum Computing (3)
- Quantum Science (11)
- Renewable Energy (1)
- Simulation (1)
- Summit (2)
- Sustainable Energy (14)
- Transformational Challenge Reactor (5)
Media Contacts
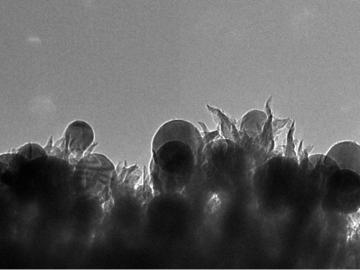
OAK RIDGE, Tenn., March 1, 2019—ReactWell, LLC, has licensed a novel waste-to-fuel technology from the Department of Energy’s Oak Ridge National Laboratory to improve energy conversion methods for cleaner, more efficient oil and gas, chemical and

Vera Bocharova at the Department of Energy’s Oak Ridge National Laboratory investigates the structure and dynamics of soft materials—polymer nanocomposites, polymer electrolytes and biological macromolecules—to advance materials and technologies for energy, medicine and other applications.
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to investigate the effectiveness of a novel crystallization method to capture carbon dioxide directly from the air.
Scientists at the Department of Energy’s Oak Ridge National Laboratory (ORNL) have developed a process that could remove CO2 from coal-burning power plant emissions in a way that is similar to how soda lime works in scuba diving rebreathers. Their research, published January 31 in...

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.

By automating the production of neptunium oxide-aluminum pellets, Oak Ridge National Laboratory scientists have eliminated a key bottleneck when producing plutonium-238 used by NASA to fuel deep space exploration.
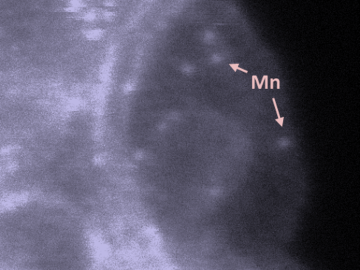
Oak Ridge National Laboratory scientists studying fuel cells as a potential alternative to internal combustion engines used sophisticated electron microscopy to investigate the benefits of replacing high-cost platinum with a lower cost, carbon-nitrogen-manganese-based catalyst.

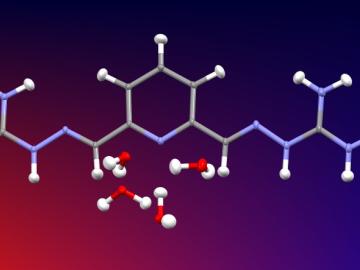
Carbon fiber composites—lightweight and strong—are great structural materials for automobiles, aircraft and other transportation vehicles. They consist of a polymer matrix, such as epoxy, into which reinforcing carbon fibers have been embedded. Because of differences in the mecha...
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.
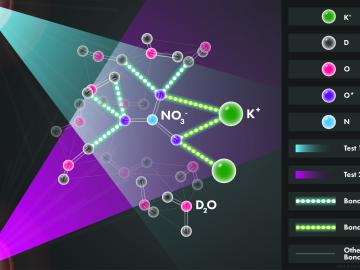
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come


