
Filter News
Area of Research
- (-) Neutron Science (105)
- (-) Transportation Systems (1)
- Advanced Manufacturing (12)
- Biological Systems (1)
- Biology and Environment (65)
- Building Technologies (2)
- Clean Energy (112)
- Climate and Environmental Systems (1)
- Computational Biology (2)
- Computational Engineering (2)
- Computer Science (9)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Functional Materials for Energy (1)
- Fusion and Fission (29)
- Fusion Energy (13)
- Isotope Development and Production (1)
- Isotopes (7)
- Materials (132)
- Materials Characterization (1)
- Materials for Computing (19)
- Materials Under Extremes (1)
- National Security (26)
- Nuclear Science and Technology (19)
- Quantum information Science (2)
- Sensors and Controls (1)
- Supercomputing (77)
News Topics
- (-) Artificial Intelligence (6)
- (-) Biomedical (11)
- (-) Fusion (1)
- (-) Materials Science (24)
- (-) Neutron Science (99)
- (-) Physics (9)
- (-) Security (2)
- (-) Sustainable Energy (2)
- 3-D Printing/Advanced Manufacturing (6)
- Advanced Reactors (1)
- Big Data (2)
- Bioenergy (6)
- Biology (5)
- Biotechnology (1)
- Chemical Sciences (2)
- Clean Water (2)
- Climate Change (1)
- Composites (1)
- Computer Science (13)
- Coronavirus (8)
- Cybersecurity (1)
- Decarbonization (3)
- Energy Storage (6)
- Environment (8)
- Fossil Energy (1)
- Frontier (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (14)
- Mathematics (1)
- Microscopy (3)
- Nanotechnology (10)
- National Security (2)
- Nuclear Energy (3)
- Polymers (1)
- Quantum Computing (1)
- Quantum Science (7)
- Space Exploration (3)
- Summit (6)
- Transportation (7)
Media Contacts
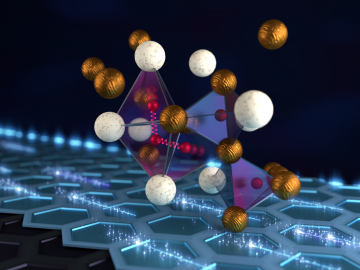
An international team of researchers has discovered the hydrogen atoms in a metal hydride material are much more tightly spaced than had been predicted for decades — a feature that could possibly facilitate superconductivity at or near room temperature and pressure.
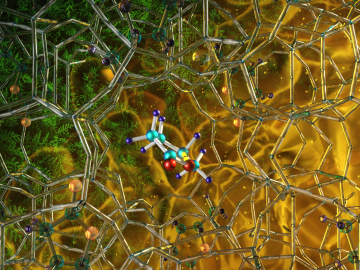
Illustration of the optimized zeolite catalyst, or NbAlS-1, which enables a highly efficient chemical reaction to create butene, a renewable source of energy, without expending high amounts of energy for the conversion. Credit: Jill Hemman, Oak Ridge National Laboratory/U.S. Dept. of Energy
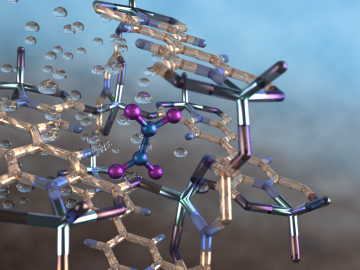
An international team of scientists, led by the University of Manchester, has developed a metal-organic framework, or MOF, material

Scientists at the U.S. Department of Energy’s Brookhaven National Laboratory have new experimental evidence and a predictive theory that solves a long-standing materials science mystery: why certain crystalline materials shrink when heated.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.
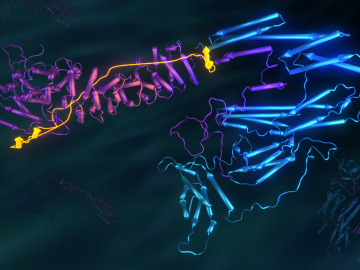
Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source and High Flux Isotope Reactor to better understand how certain cells in human tissue bond together.
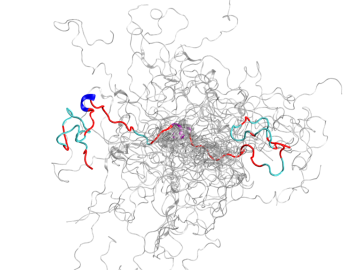
Using the Titan supercomputer and the Spallation Neutron Source at the Department of Energy’s Oak Ridge National Laboratory, scientists have created the most accurate 3D model yet of an intrinsically disordered protein, revealing the ensemble of its atomic-level structures.

Researchers at Oak Ridge National Laboratory proved that a certain class of ionic liquids, when mixed with commercially available oils, can make gears run more efficiently with less noise and better durability.

Researchers used neutron scattering at Oak Ridge National Laboratory’s Spallation Neutron Source to probe the structure of a colorful new material that may pave the way for improved sensors and vivid displays.
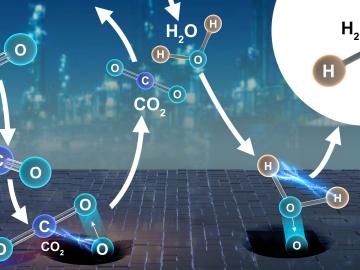
Collaborators at the Department of Energy’s Oak Ridge National Laboratory and U.S. universities used neutron scattering and other advanced characterization techniques to study how a prominent catalyst enables the “water-gas shift” reaction to purify and generate hydrogen at industrial scale.


