
Filter News
Area of Research
- Advanced Manufacturing (2)
- Biology and Environment (7)
- Clean Energy (78)
- Computer Science (2)
- Electricity and Smart Grid (1)
- Energy Sciences (1)
- Functional Materials for Energy (2)
- Fusion and Fission (10)
- Fusion Energy (7)
- Isotopes (1)
- Materials (40)
- Materials for Computing (4)
- National Security (3)
- Neutron Science (7)
- Nuclear Science and Technology (15)
- Nuclear Systems Modeling, Simulation and Validation (1)
- Supercomputing (9)
News Topics
- (-) Advanced Reactors (34)
- (-) Energy Storage (108)
- (-) Molten Salt (8)
- (-) Transformational Challenge Reactor (7)
- 3-D Printing/Advanced Manufacturing (120)
- Artificial Intelligence (91)
- Big Data (53)
- Bioenergy (91)
- Biology (98)
- Biomedical (58)
- Biotechnology (22)
- Buildings (57)
- Chemical Sciences (63)
- Clean Water (29)
- Climate Change (99)
- Composites (26)
- Computer Science (187)
- Coronavirus (46)
- Critical Materials (25)
- Cybersecurity (35)
- Decarbonization (79)
- Education (4)
- Element Discovery (1)
- Emergency (2)
- Environment (194)
- Exascale Computing (37)
- Fossil Energy (5)
- Frontier (42)
- Fusion (54)
- Grid (62)
- High-Performance Computing (84)
- Hydropower (11)
- Irradiation (3)
- Isotopes (53)
- ITER (7)
- Machine Learning (47)
- Materials (143)
- Materials Science (139)
- Mathematics (7)
- Mercury (12)
- Microelectronics (3)
- Microscopy (51)
- Nanotechnology (60)
- National Security (61)
- Net Zero (13)
- Neutron Science (131)
- Nuclear Energy (107)
- Partnerships (43)
- Physics (61)
- Polymers (33)
- Quantum Computing (34)
- Quantum Science (69)
- Renewable Energy (2)
- Security (24)
- Simulation (47)
- Software (1)
- Space Exploration (25)
- Statistics (3)
- Summit (57)
- Sustainable Energy (125)
- Transportation (97)
Media Contacts

Ask Tyler Gerczak to find a negative in working at the Department of Energy’s Oak Ridge National Laboratory, and his only complaint is the summer weather. It is not as forgiving as the summers in Pulaski, Wisconsin, his hometown.

Using additive manufacturing, scientists experimenting with tungsten at Oak Ridge National Laboratory hope to unlock new potential of the high-performance heat-transferring material used to protect components from the plasma inside a fusion reactor. Fusion requires hydrogen isotopes to reach millions of degrees.
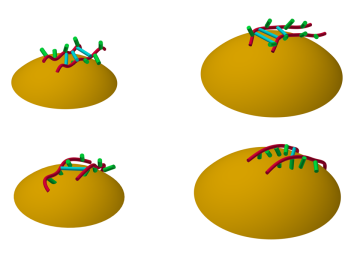
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

For the first time, Oak Ridge National Laboratory has completed testing of nuclear fuels using MiniFuel, an irradiation vehicle that allows for rapid experimentation.
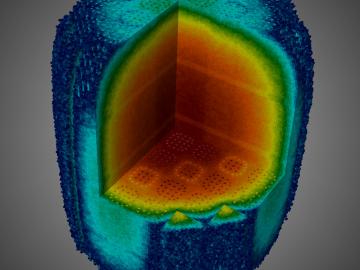
In a step toward advancing small modular nuclear reactor designs, scientists at Oak Ridge National Laboratory have run reactor simulations on ORNL supercomputer Summit with greater-than-expected computational efficiency.
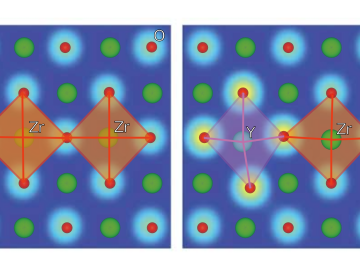
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
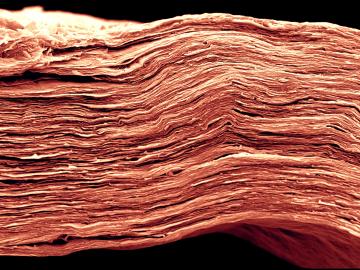

The use of lithium-ion batteries has surged in recent years, starting with electronics and expanding into many applications, including the growing electric and hybrid vehicle industry. But the technologies to optimize recycling of these batteries have not kept pace.

Oak Ridge National Laboratory scientists analyzed more than 50 years of data showing puzzlingly inconsistent trends about corrosion of structural alloys in molten salts and found one factor mattered most—salt purity.
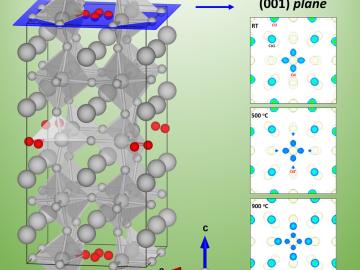
A University of South Carolina research team is investigating the oxygen reduction performance of energy conversion materials called perovskites by using neutron diffraction at Oak Ridge National Laboratory’s Spallation Neutron Source.


