
Filter News
Area of Research
News Topics
- (-) Isotopes (57)
- (-) Mercury (12)
- (-) Polymers (33)
- 3-D Printing/Advanced Manufacturing (128)
- Advanced Reactors (34)
- Artificial Intelligence (101)
- Big Data (61)
- Bioenergy (92)
- Biology (101)
- Biomedical (61)
- Biotechnology (24)
- Buildings (67)
- Chemical Sciences (73)
- Clean Water (31)
- Climate Change (106)
- Composites (30)
- Computer Science (198)
- Coronavirus (46)
- Critical Materials (29)
- Cybersecurity (35)
- Decarbonization (85)
- Education (5)
- Element Discovery (1)
- Emergency (2)
- Energy Storage (112)
- Environment (201)
- Exascale Computing (42)
- Fossil Energy (6)
- Frontier (45)
- Fusion (58)
- Grid (66)
- High-Performance Computing (93)
- Hydropower (11)
- Irradiation (3)
- ITER (7)
- Machine Learning (51)
- Materials (147)
- Materials Science (146)
- Mathematics (10)
- Microelectronics (4)
- Microscopy (51)
- Molten Salt (9)
- Nanotechnology (60)
- National Security (73)
- Net Zero (14)
- Neutron Science (137)
- Nuclear Energy (111)
- Partnerships (51)
- Physics (64)
- Quantum Computing (37)
- Quantum Science (72)
- Renewable Energy (2)
- Security (25)
- Simulation (52)
- Software (1)
- Space Exploration (25)
- Statistics (3)
- Summit (59)
- Sustainable Energy (130)
- Transformational Challenge Reactor (7)
- Transportation (99)
Media Contacts

Oak Ridge National Laboratory researchers have developed a thin film, highly conductive solid-state electrolyte made of a polymer and ceramic-based composite for lithium metal batteries.

Biological membranes, such as the “walls” of most types of living cells, primarily consist of a double layer of lipids, or “lipid bilayer,” that forms the structure, and a variety of embedded and attached proteins with highly specialized functions, including proteins that rapidly and selectively transport ions and molecules in and out of the cell.

OAK RIDGE, Tenn., Feb. 27, 2020 — Researchers at Oak Ridge National Laboratory and the University of Tennessee achieved a rare look at the inner workings of polymer self-assembly at an oil-water interface to advance materials for neuromorphic computing and bio-inspired technologies.
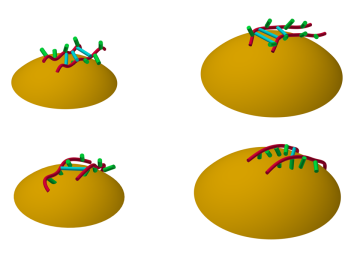
A team of researchers at Oak Ridge National Laboratory have demonstrated that designed synthetic polymers can serve as a high-performance binding material for next-generation lithium-ion batteries.

Sometimes solutions to the biggest problems can be found in the smallest details. The work of biochemist Alex Johs at Oak Ridge National Laboratory bears this out, as he focuses on understanding protein structures and molecular interactions to resolve complex global problems like the spread of mercury pollution in waterways and the food supply.

Vera Bocharova at the Department of Energy’s Oak Ridge National Laboratory investigates the structure and dynamics of soft materials—polymer nanocomposites, polymer electrolytes and biological macromolecules—to advance materials and technologies for energy, medicine and other applications.

OAK RIDGE, Tenn., Jan. 31, 2019—A new electron microscopy technique that detects the subtle changes in the weight of proteins at the nanoscale—while keeping the sample intact—could open a new pathway for deeper, more comprehensive studies of the basic building blocks of life.

Carbon fiber composites—lightweight and strong—are great structural materials for automobiles, aircraft and other transportation vehicles. They consist of a polymer matrix, such as epoxy, into which reinforcing carbon fibers have been embedded. Because of differences in the mecha...
Physicists turned to the “doubly magic” tin isotope Sn-132, colliding it with a target at Oak Ridge National Laboratory to assess its properties as it lost a neutron to become Sn-131.
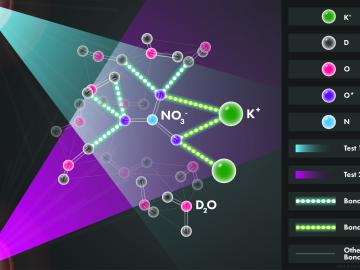
Scientists at the Department of Energy’s Oak Ridge National Laboratory used neutrons, isotopes and simulations to “see” the atomic structure of a saturated solution and found evidence supporting one of two competing hypotheses about how ions come


