
Filter News
Area of Research
- (-) Isotopes (24)
- (-) Neutron Science (19)
- (-) Quantum information Science (3)
- Advanced Manufacturing (1)
- Biological Systems (1)
- Biology and Environment (33)
- Clean Energy (61)
- Computational Biology (1)
- Computational Engineering (1)
- Computer Science (2)
- Fusion and Fission (6)
- Isotope Development and Production (1)
- Materials (53)
- Materials for Computing (8)
- National Security (19)
- Nuclear Science and Technology (5)
- Supercomputing (55)
News Type
News Topics
- (-) Biomedical (13)
- (-) Cybersecurity (2)
- (-) Energy Storage (4)
- (-) Frontier (1)
- (-) Isotopes (21)
- (-) Microscopy (4)
- (-) Space Exploration (4)
- 3-D Printing/Advanced Manufacturing (6)
- Artificial Intelligence (5)
- Big Data (2)
- Bioenergy (6)
- Biology (5)
- Biotechnology (1)
- Chemical Sciences (1)
- Clean Water (2)
- Climate Change (2)
- Composites (1)
- Computer Science (15)
- Coronavirus (8)
- Decarbonization (2)
- Environment (7)
- Fossil Energy (1)
- Fusion (1)
- High-Performance Computing (2)
- Machine Learning (3)
- Materials (14)
- Materials Science (21)
- Mathematics (1)
- Nanotechnology (9)
- National Security (3)
- Neutron Science (73)
- Nuclear Energy (5)
- Physics (8)
- Polymers (1)
- Quantum Computing (1)
- Quantum Science (10)
- Security (2)
- Summit (6)
- Sustainable Energy (3)
- Transportation (3)
Media Contacts
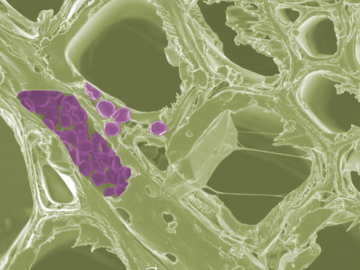
Scientists at the Department of Energy’s Oak Ridge National Laboratory have developed a new method to peer deep into the nanostructure of biomaterials without damaging the sample. This novel technique can confirm structural features in starch, a carbohydrate important in biofuel production.

Two of the researchers who share the Nobel Prize in Chemistry announced Wednesday—John B. Goodenough of the University of Texas at Austin and M. Stanley Whittingham of Binghamton University in New York—have research ties to ORNL.

Three researchers at Oak Ridge National Laboratory will lead or participate in collaborative research projects aimed at harnessing the power of quantum mechanics to advance a range of technologies including computing, fiber optics and network
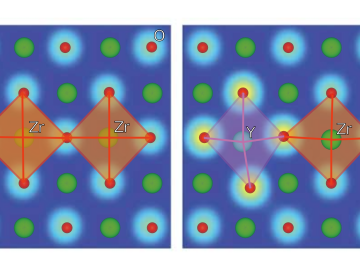
Ionic conduction involves the movement of ions from one location to another inside a material. The ions travel through point defects, which are irregularities in the otherwise consistent arrangement of atoms known as the crystal lattice. This sometimes sluggish process can limit the performance and efficiency of fuel cells, batteries, and other energy storage technologies.
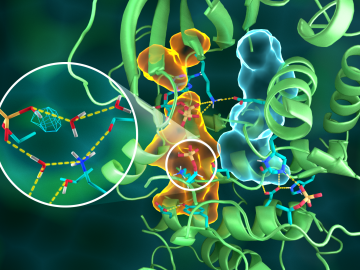
OAK RIDGE, Tenn., March 20, 2019—Direct observations of the structure and catalytic mechanism of a prototypical kinase enzyme—protein kinase A or PKA—will provide researchers and drug developers with significantly enhanced abilities to understand and treat fatal diseases and neurological disorders such as cancer, diabetes, and cystic fibrosis.


